EED is the manifestation of structural gut damage and impaired intestinal function that results from persistent gut infections in early childhood.
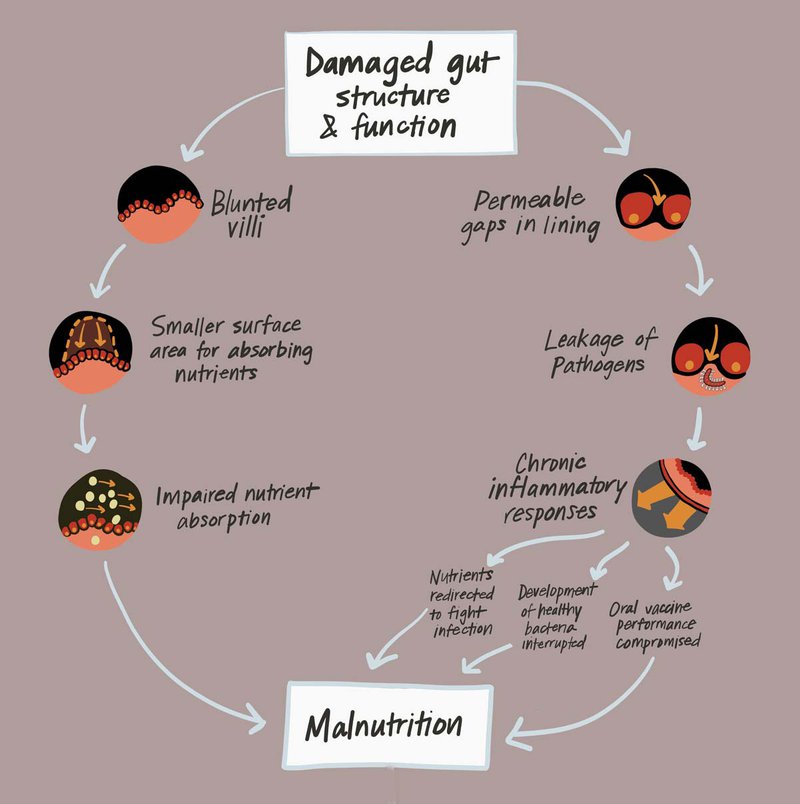
EED is virtually ubiquitous in impoverished communities, especially in areas with limited access to clean water, sanitation, and hygiene, where children are constantly exposed to an array of threatening pathogens lurking in the environment. Repeated and chronic gut infections can provoke an inflammatory response in the intestine that results in a cascade of harmful effects.
Nutrients that would typically fuel growth, cognitive development, and a strong immune system can be diverted toward fighting off infection. Chronic gut inflammation appears to cause structural changes that impede the intestine’s ability to absorb nutrients from food. It also interrupts the development of healthy commensal bacteria and compromises the performance of oral vaccines. Combined, these changes increase children’s vulnerability to further infections and risk for becoming malnourished and stunted.
Good gut health is key to optimal growth and development. Without it, children can develop environmental enteric dysfunction, which can result in chronic malnutrition and stunting. Image: PATH.
Over a lifetime, malnutrition and stunting rob children of the ability to reach their full potential. Beginning with impaired cognitive development, through poor school performance, and reduced future earnings, these conditions hold back entire communities in a cycle of poverty and poor health.
And the cycle of stunting and poor health can persist for generations. Young girls who experience growth stunting are more likely to suffer from malnutrition as adults. When undernourished women become pregnant, they are at greater risk for premature birth and low birth weight, increasing the likelihood of stunting in infants.
Stopping the cycle would mean boosting the productivity of communities and economies, and promoting global development on a massive scale.
PATH is advancing these goals by bringing together cross-sector partners to foster research, and projects aimed at enabling development of therapeutics for minimizing or improving intestinal injury.
Multi-micronutrient and EED Assessment Tool (MEEDAT)
To detect the presence of EED in individuals, we need good biomarkers, which act as an indicator of a particular disease state (think body temperature for fever). However, the current tools to quantify biomarkers of gut function and micronutrient status are expensive, time-consuming, and labor-intensive. To meet the need for an effective, affordable tool that enables detection and measurement of multiple EED biomarkers and micronutrients in low-resource settings, PATH has co-developed MEEDAT, a multiplex assay for quantifying blood-based biomarkers of EED, growth hormone resistance, systemic inflammation, and micronutrient status in children.
